
Inside the Mix – Understanding additives in 3D printed concrete
3D printed concrete offers the potential for quick sustainable construction

Rheology – from the Greek rheos - is the study of flow and is used to describe the deformation of a material under stress. The effects of rheology can be seen everywhere; compare oil, honey, and bitumen, all are liquids yet each flows very differently. Controlling rheology is crucial for the paint formulator to not only provide enhanced application properties but also to provide long term stability.
In this technical article, we will discuss the fundamental principles of rheology, how this relates to coatings, and will take a closer look at materials that can be used to modify flow behaviour.

Figure 1. Viscosities of common liquids. Viscosity depends on many variables including temperature, pressure and concentration. (Values given are estimates only)
Before we can discuss rheology we need to understand what we mean by viscosity.
Viscosity can be thought of as the internal resistance a material has to deformation. Using our earlier example, oil has a much lower viscosity than both honey and bitumen under the same conditions. Why is that?
Isaac Newton defined viscosity by imagining two infinitely large parallel plates; a lower fixed plate and a moving upper plate (Figure 2). The external force (shear stress) needed to move the top plate at a constant velocity is proportional to the velocity gradient through the liquid. This velocity gradient describes the shearing a liquid experiences i.e. the shear rate. The greater the internal friction of a fluid, the greater the amount of force required to move the top plate, and the greater the viscosity.
Molecule size and structure both play a part in viscosity; highly viscous materials consist of large, tightly linked molecules that provide high internal friction. Low viscosity fluids consist of smaller molecules with limited internal friction and therefore flow more easily. For a material to flow, it must be subjected to enough stress to overcome this internal friction (the yield point).

Figure 2. Liquids can be modelled as two plates, one moving over the other. The top plate is subjected to an external force (shear stress) and the resulting velocity the liquid experiences provides an insight into the material's viscosity.
Rheology is important not only for application properties (brushing vs spraying vs dipping) but also for many other fundamental coating characteristics such as storage stability and anti-sag properties.
On application, a paint’s viscosity must be low enough to ensure a good spreading rate and good coverage but it must also restructure quickly enough to prevent dripping and sagging. Throw into the mix the need for good levelling properties, low spattering, and in-can stability and it can be challenging to find the right rheology for a formulation (Figure 3).

Figure 3. The effect of shear rate on various coating properties. Consideration should be given to the paint not only during mixing or on application, but also at rest for long term in-can stability.
As we have already discussed liquids can have varying rheological profiles (Figure 4).
Pure substances exhibit Newtonian rheology, that is to say that their viscosity is independent of shear stress. However as coating chemists, the majority of materials we encounter do not exhibit Newtonian behaviour; most formulations are a blend of liquids and organic/inorganic particles, paint formulations therefore display much more complicated rheological profiles.
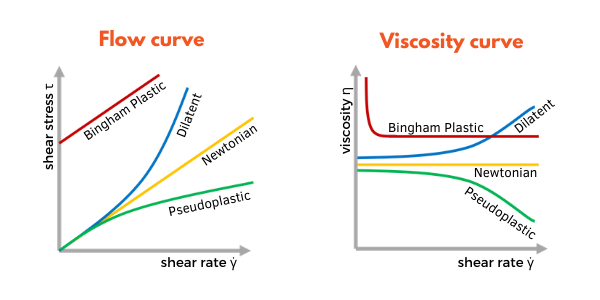
Figure 4. The relationships between shear rate γ and shear stress τ (flow) and shear rate γ and viscosity η for the different rheology profiles.
In addition to Newtonian rheology, materials can exhibit the following viscosity behaviours:
Varnishes, lacquers, and gloss paints should ideally exhibit near Newtonian rheology to enhance brushability, flow, and levelling whereas more heavily filled systems require higher viscosities at low shear to combat the effects of gravity and particle sedimentation.
In understanding a material’s rheology or flow behaviour, there are several factors we must consider:
Therefore taking into account time, temperature, and stress, measuring flow can require a rheometer rather than a viscometer. Commonly used methods of evaluating coating rheologies include:
In addition to measuring the viscosity, most formulators will also carry out tests to evaluate the levelling, sagging and spattering characteristics of their paint.
Though most of our customers will have their own viscometers and rheometers, our principal Münzing Chemie can also offer assistance with understanding the rheology of your formulation via their Technical Service.
In solventborne paints, flow properties can be regulated by the molecular weight of the dissolved binder however in waterborne formulations, rheology must be adjusted via additives (thickeners) as the binder is present as dispersed polymer particles.
There are many different chemistries that can adjust the rheology of a paint and correct thickener choice will depend on a number of factors, not only limited to the type of thickening desired, but also how and when the thickener is to be incorporated and which other additives are present in the system.
Correct thickener choice will affect colour acceptance and stability after colour addition. Other factors to consider include correct selection of dispersing agents (and correct dosage!) as this leads to an increase in solids content whilst maintaining low viscosities.
Thickeners can be broadly separated into inorganic and organic thickeners for waterborne and solventborne formulations. The below list is not exhaustive but provides an overview of various thickener types available:
Organoclays or smectites encompass a variety of naturally occurring materials and consist mostly of sodium or calcium montmorillonites and hectorite. Smectites such as bentonite are highly swellable expanding lattices which, through modification with organic alkyl groups, can be used to provide shear-thinning to aqueous and solventborne coatings.
Related to these organoclays is Attagel® (attapulgite) which is a special composite of smectite and non-expanding acicular palygorskite that forms a gel via hydrogen bonding. Unlike bentonite, Attagel can form a gel in the presence of salt and as it is highly thixotropic (Figure 5), it is especially useful for providing syneresis or liquid separation control in addition to excellent sag resistance and suspension of pigment particles.
Clays are natural materials and can be a cost effective method of controlling rheology however they have limitations, especially with regards to clear coats or applications with high-optical requirements.
 Figure 5. Left: Attapulgite under high resolution SEM. The particles are approximately 0.1 microns in diameter and 2 microns in length. Right: The effect of Attagel concentration on the viscosity of water.
Figure 5. Left: Attapulgite under high resolution SEM. The particles are approximately 0.1 microns in diameter and 2 microns in length. Right: The effect of Attagel concentration on the viscosity of water.
Pyrogenic fumed silica is widely used as an anti-settlement and rheological additive in coatings. It is produced via flame hydrolysis of silicon tetrachloride (SiCl4) and can be either untreated (hydrophilic) or surface treated with various organic groups to produce hydrophobic silica.
AEROSIL® fumed silica was the first to be produced commercially and is used in coating formulations for imparting thixotropy, anti-sag (Figure 6), and anti-settling. When properly dispersed, the silica particles form a hydrogen-bonded network which is easily broken down under shear.
In addition to improved thixotropy, selecting the correct grade of AEROSIL® also imparts benefits to the dry film including improved scratch resistance and improved water resistance and corrosion protection.
Despite low addition levels, typically 0.3 to 1.0% AEROSIL® (on total formulation), dispersion requires high shear and therefore EVONIK also now offers AERODISP® silica dispersions for easier handling and incorporation.

Figure 6. Anti-sag properties obtained by incorporating AEROSIL® fumed silica in an unsaturated polyester. Typical addition levels are 0.3 to 1.0% AEROSIL® based on total formulation.
Non-associative thickening occurs due to the entanglement of water-soluble, high molecular weight polymer chains. Formulations thickened this way display good stability and anti-settling properties as they become highly elastic. The downside however is that they often have limited flowability and the elastic properties can result in increased spattering.
Cellulose ethers are naturally sourced materials produced via reaction of cellulose with an alkali before substitution with methyl (MC), ethyl (MEC), hydroxyethyl (HEC), or hydrophobic end groups (Figure 7). This etherification process makes cellulose water-soluble and through adjusting the chain length of the cellulose backbone, a wide range of viscosity profiles can be achieved.
Cellulose ethers are often used to provide medium to high viscosities and in low to high PVC acrylic paints. As they improve water retention, they can help increase open time and offer good compatibility with other additives in the system. However, drawbacks include poorer levelling, higher spattering and poorer water resistance, and they are more prone to biological attack.
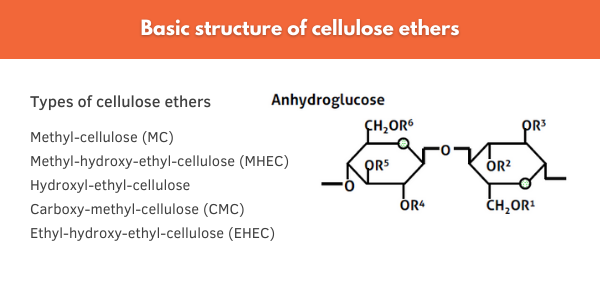
Figure 7. The basic structure of cellulose ethers. Cellulose can be modified with various hydrophobic end groups to impart thickening and water retention in aqueous formulations.
A new type of material used to impart thixotropy and control rheology is microfibrillated cellulose (also known as fibrillated cellulose, nanofibrillated cellulose, MFC or NFC). Distinct from cellulose ethers, microfibrillated celluloses like Valida have been mechanically or sometimes biologically separated out into individual fibrils. These fibrils then form an extensive network when dispersed within waterborne or polar solvents to provide excellent anti-settling and anti-sagging behaviour without a significant increase in viscosity.
In comparison to conventional thickeners like cellulose ethers or xantham gum, microfibrillated cellulose have a number of unique benefits including broad pH stability from pH 1 to 13 and high tolerance to salts whilst also providing reinforcement and enhanced open time due to their water retention properties.
Video: The use of Valida fibrillated cellulose in coatings to maximise performance
Alkali swellable polymers (ASE) are dispersions of acid-functional acrylic polymers and are particularly efficient at increasing low shear viscosity. Under acidic conditions, the acrylic polymer is tightly coiled however, on increasing the pH of the system above pH 7, these long acrylic chains become soluble in water thus thickening the solution through entanglement (Figure 8).
ASE thickeners impart elasticity into the system and are therefore a good choice for in-can stability however can result in increased spattering as a result. Care is also needed when incorporating if using a volatile alkali such as ammonia as evaporation can result in localised pH decreases and instability.

Figure 8. Alkali swellable polymers (ASE) thicken once the pH of the formulation is increased above pH 7.
Associative thickeners (Figure 9) bridge the gaps between binder molecules, pigments and fillers by creating an associative network. Hydrophobic parts on the thickener molecule associate with binder particles by adsorption at the surface or by ion-dipole interaction. The created bonds generate a given resistance against higher shear stress though can be broken down under shear. Often associative thickeners give rise to thixotropy as once shear is removed, there is a delay before the associative network rebuilds.
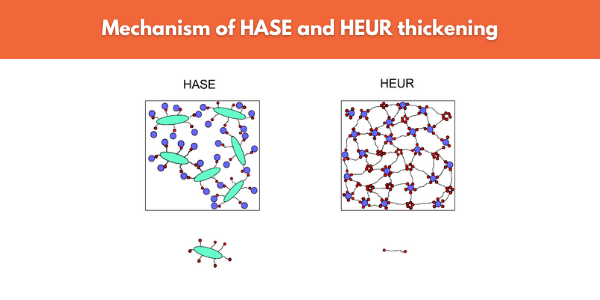 Figure 9. HASE and HEUR thickeners both associate with binder molecules (in blue) and form large polymer-thickener networks which can be broken down under shear.
Figure 9. HASE and HEUR thickeners both associate with binder molecules (in blue) and form large polymer-thickener networks which can be broken down under shear.
Like alkali swellable emulsions, HASE thickeners are acid functional acrylic polymers and are activated at pH higher than pH 7. However, in comparison to ASE thickeners, HASE have long chain hydrophobic groups attached to the polymer backbone which results in a degree of association between the thickener molecules themselves and with the binder in the formulation. Therefore increasing viscosity not just at low shear rates but also at medium and higher shear rates.
HASE thickeners such as the TAFIGEL® AP range generally allow for more Newtonian rheology compared to ASE thickeners but can also be tailored to a range of rheologies from pseudoplastic through to Newtonian.
Polyurethane thickeners or HEUR thickeners compromise of a PEG backbone with hydrophobic groups at either end. By adjusting the hydrophobic groups (length, degree of branching etc.) the type of rheology can be adjusted. Münzing’s TAFIGEL® PUR range are HEUR thickeners with rheology profiles from strongly pseudoplastic through to Newtonian behaviour.
HEUR thickeners offer very good flow and levelling and are more tolerant to pH changes than acrylic thickeners. The hydrophobic characteristics of polyurethane thickeners will also impart a degree of water resistance thus leading to improved weather resistance and wet scrub resistance. However polyurethanes are sensitive to cosolvents and surfactants and can be more difficult to formulate with.
Organo-titanates such as the TYTAN™ CX products from Borica are used to provide thixotropic structure to colloid-stabilised emulsions. Compromising of a central titanium atom and alkanolamine chelates, TYTAN™ CX will hydrogen bond to the binder particles to produce a shear reversible gel structure (Figure 10). Since some structure is maintained at medium shear, high loading of brushes or rollers are possible therefore leading to thicker wet film thicknesses
Organo-titanates are recommended to be used in combination with other thickener types such as an associative thickener or cellulosic where they give good levelling and sag resistance with low splatter.
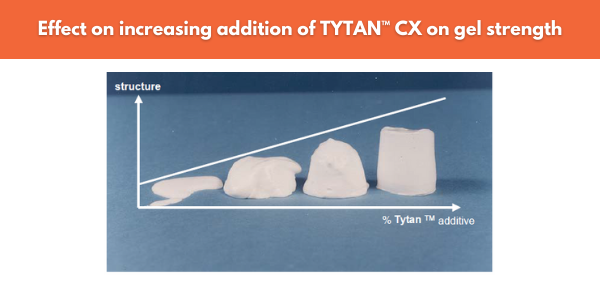 Figure 10. Effect of increasing additions of TYTAN™ CX on gel strength
Figure 10. Effect of increasing additions of TYTAN™ CX on gel strength
Understanding the rheology of your formulation is key to producing a stable paint with good application properties. There are many different additives that can be used to adjust rheology and this article serves as a brief introduction to their chemistries. In addition to the above, there are other factors that can impact on viscosity, including but not limited to: binder type, pigment/filler content, co-solvents, etc., and formulators must consider the formulation as a whole when selecting an appropriate rheology modifier.
For further insight and for recommendations for your formulation, contact your account manager or call us to discuss your requirements.
https://wiki.anton-paar.com/uk-en/basic-of-viscometry/
https://wiki.anton-paar.com/uk-en/basics-of-rheology/
https://knowledge.ulprospector.com/7491/pc-rheology-waterborne-paints/




3D printed concrete offers the potential for quick sustainable construction

Expandable graphite and Quarzwerke minerals can be used to enhance flame-retardant systems for EV safety.

High Temperature Expandable Graphite from LUH is able to be used in thermoplastics without expanding during the extrusion process.